| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website http://www.jocmr.org |
Case Report
Volume 4, Number 4, August 2012, pages 295-298
Metronidazole-Induced Neurotoxicity Developed in Liver Cirrhosis
Takatsugu Yamamotoa, b, Koichiro Abea, Hajime Anjikia, Taro Ishiia, Yasushi Kuyamaa
aDepartment of Internal Medicine, Teikyo University School of Medicine, 2-11-1 Kaga, Itabashi-ku, 173-8605, Tokyo, Japan
bCorresponding author: Takatsugu Yamamoto
Manuscript accepted for publication April 04, 2012
Short title: Metronidazole and Cirrhosis
doi: https://doi.org/10.4021/jocmr893w
| Abstract | ▴Top |
A 68 year-old-male with hepatitis C-positive liver cirrhosis was admitted because of liver abscess. After metronidazole was initiated against the infection, mental disturbance appeared. Hepatic encephalopathy was suspected at first, however, the brain MRI showed hyperintense lesion of the bilateral basal dendric nuclei which indicated metronidazole-associated encephalopathy. The symptoms became well after cessation of the drug. Metronidazole is a widely used medicine against various infections. Recent case reports describe that this medicine can induce reversible encephalopathy. However, there have been few reports regarding metronidazole-induced encephalopathy occurred in patients with cirrhosis. Here we report on a case of hepatic cirrhosis and abscess in which reversible metronidazole-induced encephalopathy developed.
Keywords: Metronidazole; Encephalopathy; Liver cirrhosis
| Introduction | ▴Top |
Metronidazole (MNZ) is a widely used medicine against various infections. Recent case reports show that this medicine can induce reversible encephalopathy [1-4]. However, there have been few reports regarding MNZ-induced encephalopathy occurred in liver cirrhosis. Here we report on a patient suffered from liver cirrhosis with recurrent hepatic encephalopathy who developed MNZ-induced neurotoxicity.
| Case Report | ▴Top |
A 68 year-old-male, who had suffered from liver cirrhosis due to hepatic C virus infection, was referred to Teikyo University hospital (Tokyo, Japan) in August 2004, because of continuous fever rising. He had several hospitalization episodes for the treatment of hepatocellular carcinoma including transarterial chemo-embolization therapy, and recurrent hepatic encephalopathy in recent five years. On admission, the body temperature reached up to 39.7 °C, the blood pressure was 105/72 mmHg, the heart rate was 95 beats/minutes, and inflammatory change and marked hepatic dysfunction was seen in the blood tests (Table 1).
 Click to view | Table 1. Laboratory Data |
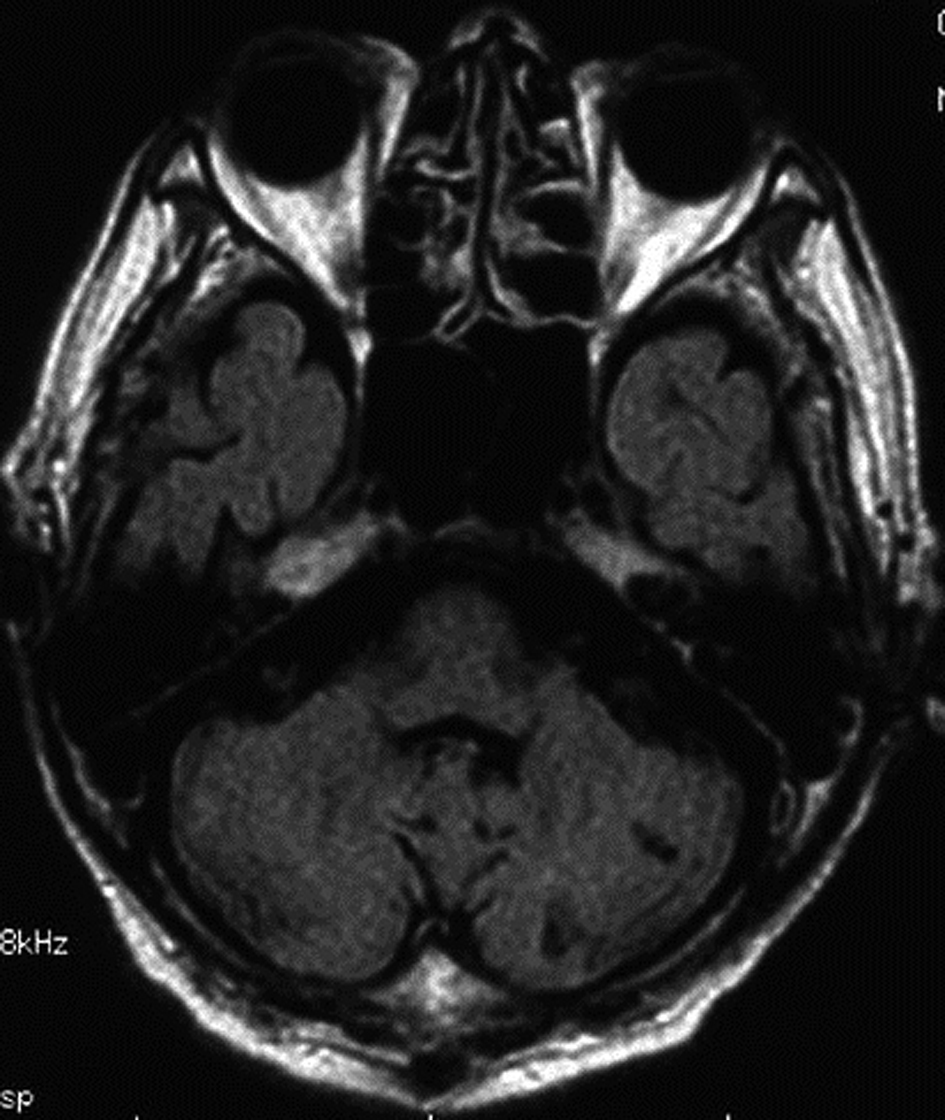
Abdominal computed tomography with contrast medium suggested the presence of liver abscess at the right lobe of the liver (Fig. 1, Fig. 2 ). Blood culture showed Klebsiella pneumonia and Clostridium perfringens, then imipenem and clindamycin was administered at first. Since the inflammatory change continued, clindamycin was changed to MNZ 1500 mg/day. Infection began to be improved after initiation of MNZ, however, speech and gait difficulty with mental disturbance suddenly appeared on the 19th day after MNZ administration. Although hepatic encephalopathy was suspected at first, the serum ammonia level did not show higher value than ever (56 mg/dL) and neurological findings were not compatible with the disease. Brain MRI showed hyperintense lesion of the bilateral basal dendric nuclei which was specific for those of metronidazole-associated encephalopathy (Fig. 3, Fig. 4), as well as hyperintensity of basal nuclei showing chronic hepatic encephalopathy (Fig. 5). After MNZ was ceased, the neurological abnormalities were improved gradually. MR image showed disappearance of hyperintensity of the dendric nuclei one month later (Fig. 6). The patient was discharged by walking alone three months later.
 Click for large image | Figure 1. Computed tomography with contrast medium (arterial phase). |
| Discussion | ▴Top |
Metronidazole is a widely used antimicrobial agent against several infections due to Clostridium difficile, amoeba, Helicobacter pylori, and so on. The serious adverse effects of the drug include bone marrow suppression, liver or renal injury, and peripheral nerve injury. Incidence of the side effect regarding central nerve system is considerably rare, however, recent case reports showed reversible neurological disorders diagnosed by specific MRI of the finding of hyperintensity at the dendric nuclei and the brain stem [1-4]. Although the pathophysiology of the phenomenon remains unclear, some have suggested the RNA binding, DNA binding of intermediate metabolites of MNZ, or the modulation of gamma-aminobutyric acid receptor as possible mechanisms [5-7]. In the present case, severe liver dysfunction due to cirrhosis would facilitate the occurrence of the side effects because MNZ is mainly metabolized by the liver [8, 9].
Diagnosis of the disease can be made by the specific MR imaging mentioned above. Regarding this patient, although past history of hepatic encephalopathy made us think the recurrence at first, the neurological findings were different from the condition. Effective treatment for MNZ-induced neurotoxicity is cessation of the drug. Although severely ill case was reported, it recovered finally. The recovery of this case seemed slower than other reported cases, probably because the liver dysfunction prolonged metabolism and excretion of the drug.
In conclusion, we experienced MNZ-induced encephalopathy developed in patients with liver cirrhosis. Brain MRI demonstrated the specific finding of the hyperintensity at the dendric nuclei, which seemed good tool for its diagnosis and evaluation of the improvement. Since patients with liver dysfunction are feasible for the adverse event of MNZ, physicians should pay attention to this possible neurological side effect.
| References | ▴Top |
- Woodruff BK, Wijdicks EF, Marshall WF. Reversible metronidazole-induced lesions of the cerebellar dentate nuclei. N Engl J Med. 2002;346(1):68-69.
pubmed - Patel K, Green-Hopkins I, Lu S, Tunkel AR. Cerebellar ataxia following prolonged use of metronidazole: case report and literature review. Int J Infect Dis. 2008;12(6):e111-114.
pubmed - Sarna JR, Brownell AK, Furtado S. Cases: Reversible cerebellar syndrome caused by metronidazole. CMAJ. 2009;181(9):611-613.
pubmed - Kim E, Na DG, Kim EY, Kim JH, Son KR, Chang KH. MR imaging of metronidazole-induced encephalopathy: lesion distribution and diffusion-weighted imaging findings. AJNR Am J Neuroradiol. 2007;28(9):1652-1658.
pubmed - Bradley WG, Karlsson IJ, Rassol CG. Metronidazole neuropathy. Br Med J. 1977;2(6087):610-611.
pubmed - Wright KH, Tyler JW. Recognizing metronidazole toxicosis in dogs. Vet Med. 2003;98:410-418.
- Evans J, Levesque D, Knowles K, Longshore R, Plummer S. Diazepam as a treatment for metronidazole toxicosis in dogs: a retrospective study of 21 cases. J Vet Intern Med. 2003;17(3):304-310.
pubmed - Loft S, Sonne J, Dossing M, Andreasen PB. Metronidazole pharmacokinetics in patients with hepatic encephalopathy. Scand J Gastroenterol. 1987;22(1):117-123.
pubmed - Cheong HC, Jeong TG, Cho YB, Yang BJ, Kim TH, Kim HC, Cho EY. Metronidazole-induced encephalopathy in a patient with liver cirrhosis. Korean J Hepatol. 2011;17(2):157-160.
pubmed
This is an open-access article distributed under the terms of the Creative Commons Attribution License, which permits unrestricted use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Clinical Medicine Research is published by Elmer Press Inc.


