| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website http://www.jocmr.org |
Case Report
Volume 8, Number 11, November 2016, pages 831-835
Bacterial Pneumonia Caused by Streptococcus pyogenes Infection: A Case Report and Review of the Literature
Nobuhiro Akuzawaa, c, Masahiko Kurabayashib
aDepartment of General Medicine, National Hospital Organization Shibukawa Medical Center, 383 Shiroi, Shibukawa, Gunma 377-0280, Japan
bDepartment of Medicine and Biological Science, Gunma University Graduate School of Medicine, 3-39-22 Showa-machi, Maebashi, Gunma 371-8511, Japan
cCorresponding Author: Nobuhiro Akuzawa, Department of General Medicine, National Hospital Organization Shibukawa Medical Center, 383 Shiroi, Shibukawa, Gunma 377-0280, Japan
Manuscript accepted for publication September 14, 2016
Short title: Pneumonia due to Streptococcus pyogenes
doi: http://dx.doi.org/10.14740/jocmr2737w
| Abstract | ▴Top |
A 78-year-old Japanese man was admitted to our hospital because of fever lasting for 4 days. His white blood cell count and C-reactive protein level were elevated and computed tomography of the chest showed bronchopneumonia in the right upper lobe of the lung. Streptococcus pyogenes was detected from sputum and blood culture samples on admission and administration of ampicillin/sulbactam was effective. Although our patient’s clinical course was good, S. pyogenes pneumonia commonly shows a high rate of fatality and septicemia, and may affect a previously healthy population. Physicians should be aware of pernicious characteristics of S. pyogenes pneumonia.
Keywords: Community-acquired pneumonia; High fatality rate; Septicemia; Streptococcus pyogenes
| Introduction | ▴Top |
Streptococcus pyogenes (S. pyogenes), or group A streptococcus, is a pathogen which causes localized illness, such as pharyngitis and skin lesions [1]. S. pyogenes can asymptomatically colonize humans, and may cause invasive diseases, such as bacteremia, pneumonia, necrotizing fasciitis, and streptococcal toxic shock syndrome [1, 2].
S. pyogenes accounts for 2-5% of cases of bacterial pneumonia in the early 20th century [3]. Fatality of pneumonia due to S. pyogenes decreased to a low level after the advent of antibiotics [4]. Sporadic outbreaks of S. pyogenes infection have been reported mainly in military camps, and S. pyogenes rarely causes pneumonia in general [5]. However, an outbreak of S. pyogenes pneumonia occurred at a US military camp in 2002 [6]. This outbreak ended after intramuscular benzathine penicillin or oral azithromycin administration, suggesting that the potential for an epidemic of S. pyogenes infection cannot be ignored.
We present here a sporadic case of S. pyogenes pneumonia. A patient was admitted to our hospital to thoroughly investigate the cause of a persistent fever of unknown origin. Chest X-ray findings were unremarkable, and plain computed tomography (CT) of the chest showed bronchopneumonia in the right upper lobe of the lung. Notably, S. pyogenes was detected from sputum and blood culture samples on admission. Our patient was discharged from our hospital after 6 g/day of aminobenzylpenicillin administration for 10 days.
| Case Report | ▴Top |
A 78-year-old Japanese man was admitted to our hospital because of a fever lasting for 4 days. He had no relevant past history, except for lung tuberculosis in his 20s, and had no family history. He had taken no medications. He had smoked 10 cigarettes a day for approximately 60 years, but did not consume alcohol. Four days before admission, he suddenly had a fever (39.4 °C) with a sore throat and chills. He went to his home doctor for a check-up and underwent an influenza antigen test, but the result was negative. Oral acetaminophen was prescribed, but his fever persisted. He visited his home doctor again 2 days before admission and had a blood test, which showed that his white blood cell (WBC) count and C-reactive protein (CRP) level were elevated (12,400/mm3 and 16.01 mg/dL, respectively.) Although oral cefcapene pivoxil (300 mg/day) was added, his fever did not improve, and he was then admitted to our hospital.
On admission, his height was 165 cm, weight was 65.8 kg, body temperature was 38.3 °C, and blood pressure was 150/75 mm Hg. His heart rate was 90 beats per minute and regular. A physical examination showed no major abnormalities, except for mild pharyngeal erythema due to pharyngitis, and mildly increased sputum excretion. His arterial blood O2 saturation (98%) and partial O2 pressure (92.1 Torr) in room air were normal. A chest X-ray film showed granular shadows that were predominantly distributed in the bilateral lower lung field (Fig. 1). An abdominal X-ray film and an electrocardiogram were normal. Laboratory tests showed a high WBC count of 11.6 × 109 cells/L (normal: 3.5 - 9.0 × 109 cells/L), lactate dehydrogenase concentration of 226 U/L (normal: 124 - 222 U/L), blood urea nitrogen concentration of 25.4 mg/dL (normal: 8 - 20 mg/dL), and CRP concentration of 16.33 mg/dL (normal: < 0.3 mg/dL). All of these variables were above the normal range. Urinary findings were normal. Rapid antigen tests for influenza, pneumococcus, and mycoplasma were negative. Therefore, ultrasonography of the abdomen was performed, but no major abnormalities were observed. Echocardiography also showed no abnormalities. Subsequently, plain CT of the chest and the abdomen was performed, and this showed bronchial wall thickening in an upper lobe of the right lung, which was not evident on a chest X-ray film (Fig. 2). Sputum culture samples for general and acid-fast bacteria and blood culture samples were collected soon after admission. He was initially diagnosed with bronchopneumonia. Intravenous administration of levofloxacin (LVFX, 500 mg/day) and ampicillin/sulbactam (ABPC/SBT, 6 g/day) was started.
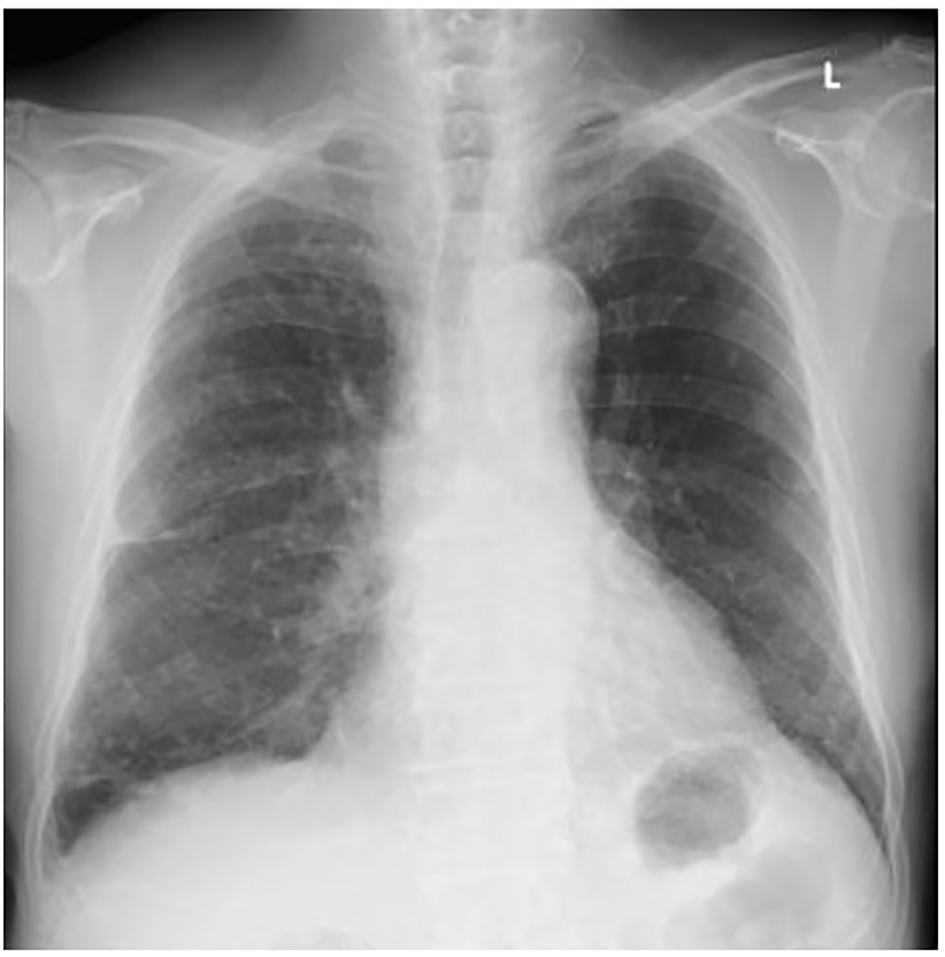 Click for large image | Figure 1. A chest X-ray film on admission. Multiple granular shadows predominantly distributed in the lower lung field, and thickening of the right interlobar pleura were observed. These findings had been pointed since the patient had had been infected with tuberculosis. No obvious findings suggesting pneumonia were observed. |
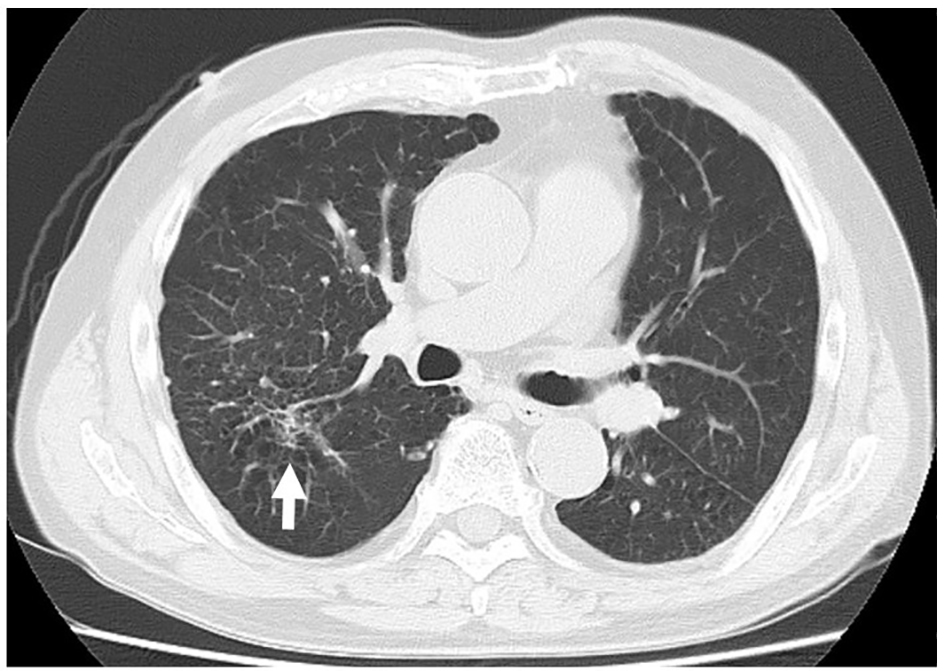 Click for large image | Figure 2. A plain computed tomography image of the lung on admission. Increased density around the lobar bronchi in the right upper lobe of the lung, which suggested bronchopneumonia (white arrow), and emphysematous changes in both lungs were observed. |
On day 2, the patient’s temperature started to fall. A sputum smear detected no obvious acid-fast bacteria. Notably, gram-positive cocci were detected in blood culture samples and these were identified as S. pyogenes on day 4. Moreover, S. pyogenes was detected from sputum culture samples. Streptococci that were detected from sputum and blood culture samples showed the same antibiotic susceptibility and were sensitive to LVFX and ABPC. On day 5, his temperature returned to normal and the WBC count was normalized (5.9 × 109 cells/L). Therefore, administration of LVFX was terminated. With regard to sputum culture, species-specific polymerase chain reaction assays for acid-fast bacteria, including Mycobacterium tuberculosis (M. tuberculosis), M. avium, M. intracellulare, and M. kansasii, were negative. On day 9, his symptoms including fever, sore throat, and increased sputum excretion, completely resolved and the CRP level returned to normal (0.19 m/dL). Therefore, oral amoxicillin (750 mg/day) was substituted for intravenous administration of LVFX and ABPC/SBT on day 10. He was discharged from our hospital on day 10 and took oral amoxicillin for 10 days. No exacerbation was observed thereafter. Image findings of bronchopneumonia disappeared on plain chest CT 1 month after discharge. The patient was lost to follow-up after this time.
| Discussion | ▴Top |
Pneumonia caused by S. pyogenes is an uncommon cause of community-acquired pneumonia, but it may show a high mortality rate. Barnham et al [7] reviewed 17 cases of S. pyogenes pneumonia and reported its high mortality rate (47%), as well as a high detection rate (88%) of S. pyogenes from blood culture samples. They reported that 63% of patients who did not survive died within 1 day after hospitalization [7]. A recent study also showed a high mortality rate (20%) of S. pyogenes pneumonia, with half of the patients dying within 24 h after admission, and the detection rate of S. pyogenes from blood culture was 43% (17/40 cases) [8]. S. pyogenes pneumonia commonly occurs in winter and spring [7, 8]. Hypertension, chronic obstructive pulmonary disease, and diabetes have been reported as frequent comorbidities in patients with S. pyogenes pneumonia [7, 8]. With regard to complications of S. pyogenes pneumonia, pleural effusion, cavity formation in the lungs, septicemia, and septic shock have been reported [7, 8]. Symptoms mimicking scarlet fever, including desquamating rash, may be observed in rare cases [7]. Despite these serious conditions, there have been few other studies on the clinical presentation, prognosis, and characteristics of S. pyogenes pneumonia. To the best of our knowledge, 16 case reports describing patients with S. pyogenes pneumonia, including our case, have been published in the past three decades (Table 1) [9-22]. The duration from onset of S. pyogenes pneumonia to hospitalization varied from 1 to 9 days, and the mean duration was 4.8 ± 2.4 days. Among these 16 cases, six (38%) patients died after hospitalization [9, 13, 16, 19-21] and four of the six patients who did not survive died within 1 day after hospitalization [9, 16, 19, 20]. Blood culture samples were positive for S. pyogenes in 12 (75%) patients. With regard to the background of these patients, three had a history of respiratory illness (asthma, chronic obstructive pulmonary disease, and obsolete pulmonary tuberculosis) [10, 16], one underwent anti-tumor necrosis factor therapy using adalimumab for rheumatoid arthritis [20], one was an intravenous drug abuser [11], and one underwent thyroidectomy for treatment of thyroid cancer 5 days before onset of S. pyogenes pneumonia [18]. However, apart from these six patients having underlying diseases or risk factors of immunodeficiency, the remaining 10 (63%) patients were previously healthy. Moreover, based on duration from onset to hospitalization, the patients could be divided into two groups: 1) ≤ 5-day group (shorter than average duration, nine patients) and 2) > 5-day group (longer than average duration, seven patients). In the ≤ 5-day group, three of nine (33%) patients died after hospitalization [9, 16, 21], while three of seven (43%) patients died in the > 5-day group [13, 19, 20]. Interestingly, in the ≤ 5-day group, three patients showed co-infection of influenza B virus and S. pyogenes, and one of these three patients died [21, 22]. However, in the > 5-day group, there was only one case of influenza A virus (IAV) infection. Acute viral tract infections can be associated with the occurrence of S. pyogenes pneumonia, reflecting virus-induced epithelial damage in the respiratory tract together with transient immune suppression [7]. Tamayo et al [8] also suggested the potential of influenza B virus for aggravation of S. pyogenes pneumonia. Therefore, physicians should be careful about preceding influenza virus infection in patients with S. pyogenes pneumonia.
 Click to view | Table 1. Sixteen Adult Cases of S. pyogenes Pneumonia Reported in the Past Three Decades |
Previous studies have shown that IAV infection facilitates S. pyogenes infection in the respiratory tract. IAV infection increases the abundance of fibrinogen 4 days after infection [1]. Fibrinogen is important for S. pyogenes adherence and internalization within epithelial cells via M protein, which is adhesion molecule on the streptococcal surface and triggers uptake of bacteria into the host cells [23]. Production of fibronectin, another important ligand for S. pyogenes, also increases during IAV infection [1]. On the other hand, viral hemagglutinin and neuraminidase also stimulate expression of adhesion molecules on the cell surface [1]. These findings suggest that IAV infection may promote S. pyogenes infection in the respiratory tract. The outcome of IAV and S. pyogenes co-infections in murine models varies depending on the specific viral and bacterial strains [1]. However, whether influenza B virus more rapidly deteriorates S. pyogenes infection than IAV is unclear because of a lack of adequate comparative studies. In addition, limitations of this report include our inability to determine the M protein serotype of the S. pyogenes strain detected from our patient. Additionally, we were unable to show the genetic identity of the S. pyogenes strains obtained from sputum and blood culture samples. Further research to determine other factors, including preceding viral infection other than influenza and mechanisms exacerbating S. pyogenes pneumonia, is required.
In conclusion, we present a case of S. pyogenes pneumonia. Although our patient’s clinical course was good, our literature review showed a high mortality rate of S. pyogenes pneumonia (38%). Notably, 63% of the reviewed patients presenting with S. pyogenes pneumonia were previously healthy. A high detection rate (75%) of S. pyogenes from blood culture samples is also a characteristic in S. pyogenes pneumonia. Although S. pyogenes is not a major pathogen of community-acquired pneumonia, physicians should be aware of its characteristics.
Grant Support
No support was received specifically for this work.
Funding
This research received no specific grant from any funding agency in the public, commercial, or not-for-profit sectors.
Conflicts of Interest
The authors declare that they have no conflicts of interest.
| References | ▴Top |
- Herrera AL, Huber VC, Chaussee MS. The Association between Invasive Group A Streptococcal Diseases and Viral Respiratory Tract Infections. Front Microbiol. 2016;7:342.
doi pubmed - O'Loughlin RE, Roberson A, Cieslak PR, Lynfield R, Gershman K, Craig A, Albanese BA, et al. The epidemiology of invasive group A streptococcal infection and potential vaccine implications: United States, 2000-2004. Clin Infect Dis. 2007;45(7):853-862.
doi pubmed - Keefer CS, Rantz LA, Rammelkamp CH. Hemolytic Streptococcal Pneumonia and Empyema: A Study of 55 Cases with Special Reference to Treatment. Trans Am Clin Climatol Assoc. 1940;56:31-56.
pubmed - Basiliere JL, Bistrong HW, Spence WF. Streptococcal pneumonia. Recent outbreaks in military recruit populations. Am J Med. 1968;44(4):580-589.
doi - Welch CC, Tombridge TL, Baker WJ, Kinney RJ. betahemolytic streptococcal pneumonia: report of an outbreak in a military population. Am J Med Sci. 1961;242:157-165.
doi pubmed - Crum NF, Russell KL, Kaplan EL, Wallace MR, Wu J, Ashtari P, Morris DJ, et al. Pneumonia outbreak associated with group a Streptococcus species at a military training facility. Clin Infect Dis. 2005;40(4):511-518.
doi pubmed - Barnham M, Weightman N, Anderson A, Pagan F, Chapman S. Review of 17 cases of pneumonia caused by Streptococcus pyogenes. Eur J Clin Microbiol Infect Dis. 1999;18(7):506-509.
doi pubmed - Tamayo E, Montes M, Vicente D, Perez-Trallero E. Streptococcus pyogenes Pneumonia in Adults: Clinical Presentation and Molecular Characterization of Isolates 2006-2015. PLoS One. 2016;11(3):e0152640.
doi pubmed - McMurray JJ, Fraser DM, Brogan O. Fatal Streptococcus pyogenes pneumonia. J R Soc Med. 1987;80(8):525-526.
pubmed - McIntyre HD, Armstrong JG, Mitchell CA. Streptococcus pyogenes pneumonia with abscess formation. Aust N Z J Med. 1989;19(3):248-249.
doi pubmed - McWhinney PH, Nathwani D. Streptococcus group A pneumonia in an intravenous drug misuser (IVDM). Eur Respir J. 1991;4(6):761-763.
pubmed - Hamour A, Bonnington A, Wilkins EG. Severe community acquired pneumonia associated with a desquamating rash due to group A beta-haemolytic streptococcus. J Infect. 1994;29(1):77-81.
doi - Case records of the Massachusetts General Hospital. Weekly clinicopathological exercises. Case 13-1996. A 51-year-old man with the adult respiratory distress syndrome. N Engl J Med. 1996;334(17):1116-1123.
doi pubmed - Kalima P, Riordan T, Berrisford RG, Sarsfield PT. Necrotizing pneumonia associated with group A streptococcal bacteraemia. Eur J Clin Microbiol Infect Dis. 1998;17(4):296-298.
doi pubmed - Birch C, Gowardman J. Streptoccocus pyogenes: a forgotten cause of severe community-acquired pneumonia. Anaesth Intensive Care. 2000;28(1):87-90.
pubmed - Taylor MB, Barkham T. Fatal case of pneumonia caused by a nonhemolytic strain of Streptococcus pyogenes. J Clin Microbiol. 2002;40(6):2311-2312.
doi - Papadas TA, Karkoulias KP, Mastronikolis NS, Sampsonas FL, Spiropoulos KB. Concurrent tonsillitis and pneumonia due to S. pyogenes. Clin Ter. 2008;159(1):33-34.
pubmed - Saldias F, Yanez J, Saldias V, Diaz O. [Community-acquired pneumonia caused by Streptococcus pyogenes: report of one case]. Rev Med Chil. 2008;136(12):1564-1569.
doi pubmed - Izumiyama N, Miki H, Shishikura Y, Kawaguchi C, Saitou W, Kikuchi T, Kumagai K, et al. [Case of severe streptococcus pyogenes pneumonia with streptococcus toxic shock syndrome]. Nihon Kokyuki Gakkai Zasshi. 2008;46(6):488-492.
pubmed - Weinblatt ME, Abbott GF, Koreishi AF. Case records of the Massachusetts General Hospital. Case 13-2009. A 54-year-old woman with respiratory failure and a cavitary lesion in the lung. N Engl J Med. 2009;360(17):1770-1779.
doi pubmed - Aebi T, Weisser M, Bucher E, Hirsch HH, Marsch S, Siegemund M. Co-infection of Influenza B and Streptococci causing severe pneumonia and septic shock in healthy women. BMC Infect Dis. 2010;10:308.
doi pubmed - Lam KW, Sin KC, Au SY, Yung SK. Uncommon cause of severe pneumonia: co-infection of influenza B and Streptococcus. Hong Kong Med J. 2013;19(6):545-548.
doi pubmed - Rohde M, Cleary PP. Adhesion and invasion of streotococcus pyogenes into host cells and clinical relevance of intracellular streptococci. In: Streptococcus pyogenes: basic biology to clinical manifestations [internet]. Ferretti JJ, Stevens DL, Fischetti VA, Eds. Oklahoma City (OK): University of Oklahoma Health Science Center, 2016. Available at http://www.ncbi.nlm.nih.gov/books/NBK333424/.
This is an open-access article distributed under the terms of the Creative Commons Attribution-NonCommercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Clinical Medicine Research is published by Elmer Press Inc.


