| Journal of Clinical Medicine Research, ISSN 1918-3003 print, 1918-3011 online, Open Access |
| Article copyright, the authors; Journal compilation copyright, J Clin Med Res and Elmer Press Inc |
| Journal website http://www.jocmr.org |
Original Article
Volume 9, Number 9, September 2017, pages 782-787
Serum Betatrophin Levels and Clinical Features in Patients With Poorly Controlled Type 2 Diabetes
Kohzo Takebayashia, b, Kenji Haraa, Tomoko Terasawaa, Rika Narusea, Mariko Suetsugua, Takafumi Tsuchiyaa, Toshihiko Inukaia
aDepartment of Internal Medicine, Dokkyo Medical University Koshigaya Hospital, Koshigaya, Saitama, Japan
bCorresponding Author: Kohzo Takebayashi, Department of Internal Medicine, Dokkyo Medical University Koshigaya Hospital, 2-1-50, Minami-Koshigaya, Koshigaya, Saitama 343-8555, Japan
Manuscript submitted June 22, 2017, accepted July 7, 2017
Short title: Serum Betatrophin Levels and Clinical Features
doi: https://doi.org/10.14740/jocmr3114w
| Abstract | ▴Top |
Background: Betatrophin is a hormone mainly secreted by the liver that influences lipid metabolisms. The main purposes of this study were to investigate the effect of canagliflozin (a sodium glucose transporter 2 inhibitor) on circulating betatrophin levels, and to investigate the correlation of various markers associated with glucose and lipid metabolisms with betatrophin in patients with poorly controlled type 2 diabetes.
Methods: Patients were randomly divided into a control group (n = 15) and a canagliflozin-treated group (n = 15). After hospitalization, the canagliflozin-treated group took 100 mg/day of canagliflozin for 3 days. Blood tests were performed at baseline and after 3 days of treatment.
Results: Canagliflozin treatment for 3 days did not significantly change fasting and postprandial serum betatrophin levels. On the other hand, betatrophin levels had a significant positive correlation with hemoglobin A1c, fasting plasma glucose, and high-density lipoprotein cholesterol levels at baseline.
Conclusions: The current study suggests that short-term treatment by canagliflozin does not influence circulating betatrophin levels, and that betatrophin is positively associated with markers of glycemic control and high-density lipoprotein cholesterol in patients with poorly controlled type 2 diabetes.
Keywords: Betatrophin; Canagliflozin; Type 2 diabetes
| Introduction | ▴Top |
Betatrophin, also known as an angiopoietin-like protein 8 (ANGPTL8) or lipasin, is a hormone produced mostly in liver in human [1-3]. A previously reported effect of betatrophin on regeneration of pancreatic β cells is currently considered to be negative [4-8]. However, some recent studies show positive association of circulating betatrophin levels with markers of glycemic control in patients with type 2 diabetes [9-11], although the association is not investigated fully in poorly controlled type 2 diabetes with the mean hemoglobin (Hb) A1c levels over 10%. The mechanisms and clinical significances of potentially these associations are yet not apparent fully.
On the other hand, recent clinical studies have also demonstrated the positive associations of circulating betratophin levels with serum lipids levels in patients with impaired glucose tolerance (IGT) or type 2 diabetes [11, 12], and in healthy subjects [11-13]. Since it is suggested that betatrophin is associated with circulating low-density lipoprotein cholesterol (LDL-C), high-density lipoprotein cholesterol (HDL-C), and triglyceride (TG) probably via the respectively different mechanisms [1], it is likely that the change of circulating betatrophin levels by the treatment influences these serum lipids levels. The production of betatrophin in the liver is greatly enhanced under insulin resistance caused by the administration of an insulin receptor blocker [4], and therefore it may be theologically possible that the agents which can decrease circulating insulin levels, such as sodium glucose transporter 2 (SGLT2) inhibitors [14], increase circulating betatrophin levels, resulting in the change of serum lipids levels. However, it is still unclear whether these agents can influence circulating betatrophin levels in clinical practice.
In the current study, we studied the effect of canagliflozin (an SGLT2 inhibitor) on circulating betatrophin levels in patients with poorly controlled type 2 diabetes by the randomized open-label study design. We also investigated the correlation of betatrophin levels with the various circulating markers associated with glucose and lipid metabolisms at baseline in these patients. Before performing the study, we hypothesized that canagliflozin significantly may increase circulating betatrophin levels, and that betatrophin would positively associated with the markers of glycemic control and serum lipids levels even under the condition of poor glycemic control as this study.
| Patients and Methods | ▴Top |
Patients
The study, which was named CANARIA (the effect of CANAgliflozin on active GLP-1 and betatRophin in patients with type 2 dIAbetes), was performed from February 2015 to December 2015 at Dokkyo Medical University Koshigaya Hospital (Koshigaya, Japan). Although the original protocol included the measurement of the glucagon-like peptide (GLP)-1, which is a hormone that stimulates the secretion of insulin in pancreatic β cells, we have focused on the effect of canagliflozin on serum betatrophin levels and on the correlation of betatrophin with various markers of glucose and lipid metabolisms in this paper. This study was registered at UMIN000016539 (https://upload.umin.ac.jp/cgi-openbin/ctr/ctr.cgi?function=brows&action=brows&type=summary&recptno=R000019197&language=E).
The detailed study protocol has been already presented [15, 16]. In brief, this study was a randomized open-label study. Key study inclusion criteria included: 1) patients (male or female) aged 20 or over at enrollment, 2) patients diagnosed as having type 2 diabetes (based on the criteria of the Japan Diabetes Society 2010) [17], 3) patients in whom the type and dose of all anti-diabetic drugs was not changed during 4 weeks before starting the study, 4) patients who had not taken dipeptidyl peptidase-4 (DPP4) inhibitors, GLP-1 receptor agonists, and SGLT2 inhibitors during the 1 month before starting the study, and 5) patients who are hospitalized for glycemic control for type 2 diabetes. Key study exclusion criteria included: 1) patients with type 1 diabetes, pancreatic diabetes, or secondary diabetes, diabetic coma or diabetic pre-coma, 2) patients who are not candidates for canagliflozin treatment, 3) patients under insulin treatment, and 4) patients with severe renal dysfunction (serum creatinine (Cr) > 2.4 mg/dL or creatinine clearance (Ccr) < 30 mL/min). The initial target number of recruited patients was 40, and during the study period, 39 patients were enrolled. The patients were randomly assigned to two groups (canagliflozin-treated group (n = 19) and control group (n = 20)), by a central computer system based on HbA1c, body mass index (BMI), gender, and age. Four patients in the control group and two in the canagliflozin-treated group voluntarily withdrew from the study before initiation of the first meal test in the study. One patient in the canagliflozin-treated group was released from the study because the patient had had type 1 diabetes on admission. One patient in the control group and one in canagliflozin-treated group were excluded because they had received co-treatment with a DPP4 inhibitor just before hospitalization. As a result, 15 patients in the control group and 15 in the canagliflozin-treated group finished the study.
The characteristics of the entire 30 (20 males and 10 females) patients have been previously presented in detail [16]. In brief, all patients’ mean age, BMI, HbA1c, and fasting plasma glucose (FPG) were 58.7 ± 11.4 years, 25.5 ± 5.0 kg/m2, 10.4±2.3%, and 181.0 ± 65.6 mg/dL, respectively. There were no significant differences in age, BMI, HbA1c and FPG between control and canagliflozin-treated groups.
Methods
On day 2 (the next day after hospitalization), patients in the canagliflozin-treated group took canagliflozin (100 mg, one tablet) after the first meal test (before lunch). Thereafter, patients in this group took canagliflozin 30 min before eating breakfast (test meal) in all days since next day (day 3). Bloods for betatrophin were collected at 0 (just before eating), and at 120 min on the day of the meal tests (i.e., days 2 and 5). The reason for the relatively short-term observation periods (3 days) in the current study was mainly due to the ethical considerations because the patients were those who were hospitalized for glycemic control as above described, and because the patients in the control group did not receive additional anti-diabetic drugs during this period.
Meal test
The commercially available test meal, meal test C (cookie type) (Saraya Co., Ltd, Osaka, Japan) was used. Each patient took one container of meal test C basically at 9:00 after overnight fasting for at least 10 h (from 20:00 the previous day) on days 2 and 5 after hospitalization. The cookie (one container) consisted of 75 g of carbohydrate (0 g of sucrose), 28.5 g of fat, 8.0 g of protein, 0.5 - 4.0 g of dietary fiber, and 125 mg of sodium, yielding 592 kcal as total calories. Patients were required to take all test meals with some water at least within 15 min of the test.
Measurement of serum betatrophin
Serum betatrophin levels were measured in duplicate using a human betatrophin EIA kit (Phoenix Pharmaceuticals, Inc., Burlingame, CA). Based on the manufacturer’s information, the intra-assay and inter-assay coefficient of variability (CV) values were less than 10% and less than 15%, respectively [18].
Ethical considerations
All subjects provided written informed consent for inclusion in the study. The study was approved by the Local Ethics Committee at our hospital and was performed according to the guidelines of the Declaration of Helsinki.
Statistical analysis
Comparisons between the two groups for age, BMI, HbA1c and FPG were made using an unpaired t-test. The two time points for betatrophin in control or canagliflozin-treated group were compared using a paired t-test. In the correlation analysis of betatrophin with multiple variables, and multiple regression analysis with betatrophin as the dependent variable, these variables excluding aspartate transaminase (AST), alanine transaminase (ALT), gamma-glutamyl transpeptidase (GGT) and high-sensitivity C-reactive protein (hsCRP) followed a normal distribution, and the AST, ALT, GGT and hsCRP were log10-transformed because of the skewed distributions. Multiple regression analysis was performed by using the stepwise forward selection method. All statistical analyses were performed using Ekuseru-Toukei 2012 software (Social Survey Research Information Co., Ltd, Tokyo, Japan). A P value of less than 0.05 was accepted as indicating statistical significance (two-sided).
| Results | ▴Top |
The results for the change on various markers including glucose, insulin and lipids levels by canagliflozin-treatment for 3 days have been previously presented in detail [15]. The mean values of fasting and postprandial betatrophin in all 30 patients were 0.29 ± 0.21 and 0.25 ± 0.18 ng/mL, respectively. The change in fasting and in postprandial betatrophin levels between day 2 and day 5 in both the control and canagliflozin-treated groups is shown in Figure 1. Although a tendency toward higher levels of fasting and postprandial betatrophin at baseline in the control group compared with the canagliflozin-treated group was found, there were no statistical differences in these betatrophin levels between the control and the canagliflozin-treated groups: 0.36 ± 0.26 vs. 0.22 ± 0.12 ng/mL in fasting serum betatrophin levels (95% confidential interval for the difference: -0.01 to 0.29, P = 0.0688), and 0.30 ± 0.22 vs. 0.20 ± 0.12 ng/mL in postprandial serum betatrophin levels (95% confidential interval for the difference: -0.04 to 0.23, P = 0.1475). The meal test did not significantly change serum betatrophin levels in both groups (Fig. 1a, b). Canagliflozin-treatment did not have an effect on both fasting and postprandial serum betatrophin levels (Fig. 1b). The correlations of fasting serum betatrophin levels with multiple variables, and the results of stepwise analysis with fasting serum betatrophin levels as the dependent variables at baseline in all patients are shown in Table 1. There were significant positive correlations of betatrophin levels with HbA1c, FPG and HDL-C levels. Betatrophin levels also had significant positive association with HbA1c and HDL-C levels in multiple regression analysis. The correlation of HbA1c and HDL-C levels with betatrophin levels is shown also in Figure 2.
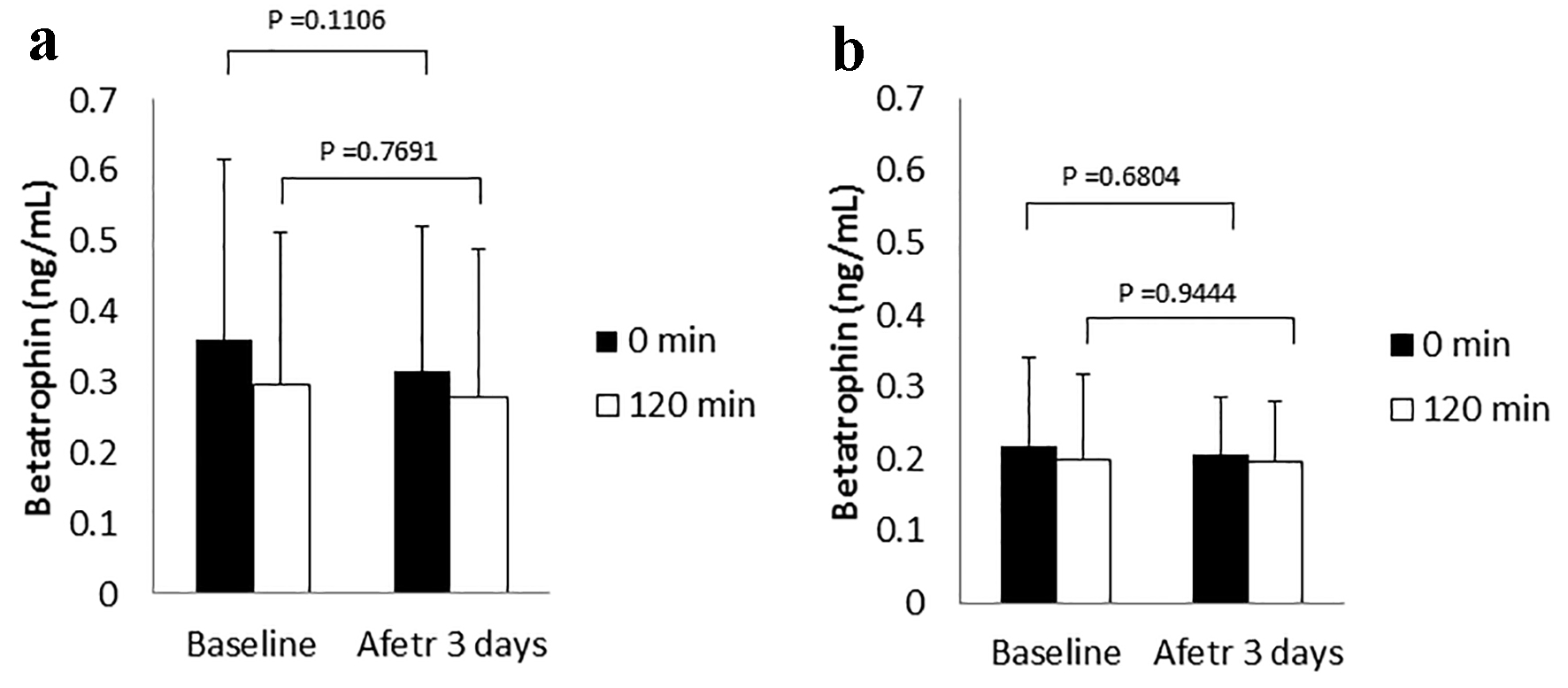 Click for large image | Figure 1. Betatrophin levels in the control (a) and canagliflozin-treated groups (b). |
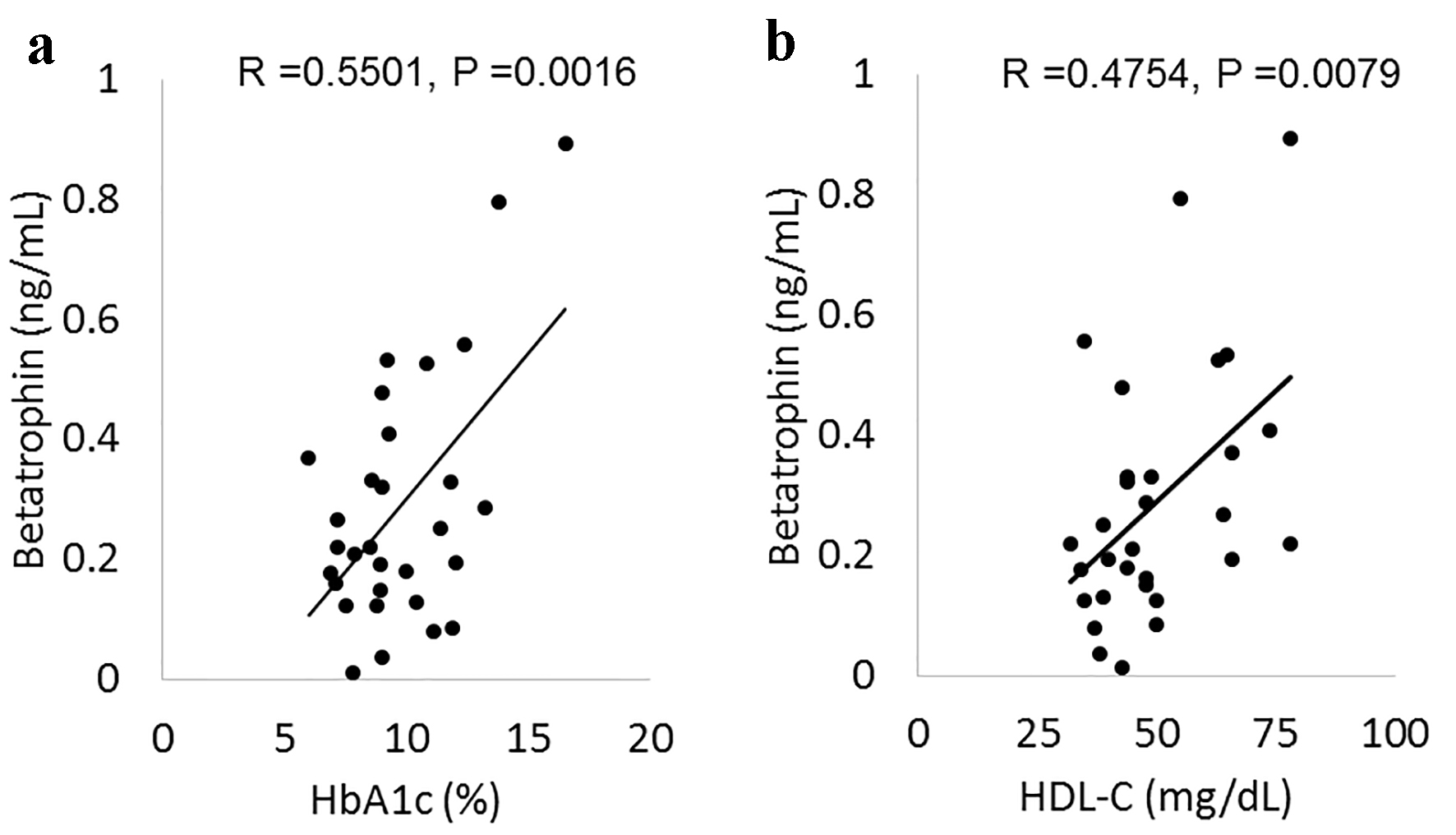 Click for large image | Figure 2. The correlation of betatrophin with HbA1c (a) and HDL-C (b). HbA1c: hemoglobin A1c; HDL-C: high-density lipoprotein cholesterol. |
 Click to view | Table 1. The Correlation of Betatrophin With Multiple Variables, and Stepwise Regression Analysis With Betatrophin as the Dependent Variable |
| Discussion | ▴Top |
In the current study, we investigated the effect of canagliflozin (an SGLT2 inhibitor) on serum betatrophin levels. Because SGLT2 inhibitors decrease insulin levels [14], and because circulating betatrophin levels were remarkably elevated by administration of an insulin receptor blocker resulting in liver insulin resistance [4], we hypothesized that administration of SGLT2 inhibitors, such as canagliflozin, may increase circulating betatrophin levels and therefore may influence serum lipid concentrations. Contrary to our expectations, canagliflozin-treatment did not change both fasting and postprandial serum betatrophin levels, despite the fact that this agent clearly decreased serum insulin levels also in the current study [15]. At the present time, the reason for the result is unknown. However, the result in the current study was that in a short-term observation, therefore it may be possible that this influenced the result at least partially.
Betatrophin levels were not influenced by the meal test in the current study. This finding appears to support the result in a previous animal study where the production of betatrophin in the liver did not change with a normal diet [2], because betatrophin is mainly produced in the liver in humans [3]. In the current study, there was a significant positive correlation of betatrophin with HbA1c and FPG, corresponding with the results of some previous studies in patients with IGT or type 2 diabetes [9-11]. These associations appear to be robust because the positive correlations were evident even in the patients in this study who had mostly poorer glycemic control as reflected by their HbA1c levels (approximately 10%), compared with the previous studies [9-11]. The reason for the potentially positive association between betatrophin and glycemic control as evaluated by HbA1c or FPG remains unclear since the effect of betatrophin on regeneration of pancreatic β cells which was previously reported [4] is currently considered to be negative [5-8].
Betatrophin also had a significant positive correlation with HDL-C in accordance with the results of some previous studies [11, 12], although these associations were not found in other reports [19]. This positive association may be reasonable because it is suggested that betatrophin may be an important regulator of HDL-C [20]. On the other hand, in the current study, betatrophin did not correlate with LDL-C and TG, whose positive associations with betatrophin have been also reported [19]. It should be noted that the mechanisms for these associations may be respectively different although the detailed mechanisms are not yet apparent fully [1]. Speculatively it may be possible that poor glycemic control in the patients in this study influenced the potential associations of LDL-C or TG levels with betatrophin levels. Our results might suggest that the treatment of elevating betatrophin may be beneficial in the aspect of increasing HDL-C, which is generally considered to possess anti-atherogenic effects [21], especially in patients with poorly controlled type 2 diabetes.
In the current study, the numbers of the patients in both the control and the canagliflozin-treated groups were small, and the treatment time was for merely 3 days, which are the limitations of this study.
In conclusion, in patients with poorly controlled type 2 diabetes, canagliflozin-treatment for 3 days did not significantly change fasting and postprandial serum betatrophin levels. Circulating betatrophin levels had a positive correlation with HbA1c, FPG and HDL-C levels in these patients. For understanding these findings, a future study with larger numbers of patients and longer observation period is needed.
Funding Support
This study was supported by funds for institution from Mitsubishi Tanabe Pharma Co. (Osaka, Japan).
Conflicts of Interest
The authors have no conflicts of interest to disclose.
| References | ▴Top |
- Quagliarini F, Wang Y, Kozlitina J, Grishin NV, Hyde R, Boerwinkle E, Valenzuela DM, et al. Atypical angiopoietin-like protein that regulates ANGPTL3. Proc Natl Acad Sci U S A. 2012;109(48):19751-19756.
doi pubmed - Zhang R. Lipasin, a novel nutritionally-regulated liver-enriched factor that regulates serum triglyceride levels. Biochem Biophys Res Commun. 2012;424(4):786-792.
doi pubmed - Ren G, Kim JY, Smas CM. Identification of RIFL, a novel adipocyte-enriched insulin target gene with a role in lipid metabolism. Am J Physiol Endocrinol Metab. 2012;303(3):E334-351.
doi pubmed - Yi P, Park JS, Melton DA. Betatrophin: a hormone that controls pancreatic beta cell proliferation. Cell. 2013;153(4):747-758.
doi pubmed - Wang Y, Quagliarini F, Gusarova V, Gromada J, Valenzuela DM, Cohen JC, Hobbs HH. Mice lacking ANGPTL8 (Betatrophin) manifest disrupted triglyceride metabolism without impaired glucose homeostasis. Proc Natl Acad Sci U S A. 2013;110(40):16109-16114.
doi pubmed - Jiao Y, Le Lay J, Yu M, Naji A, Kaestner KH. Elevated mouse hepatic betatrophin expression does not increase human beta-cell replication in the transplant setting. Diabetes. 2014;63(4):1283-1288.
doi pubmed - Gusarova V, Alexa CA, Na E, Stevis PE, Xin Y, Bonner-Weir S, Cohen JC, et al. ANGPTL8/betatrophin does not control pancreatic beta cell expansion. Cell. 2014;159(3):691-696.
doi pubmed - Yi P, Park JS, Melton DA. Retraction Notice to: Betatrophin: A Hormone that Controls Pancreatic beta Cell Proliferation. Cell. 2017;168(1-2):326.
doi pubmed - Espes D, Martinell M, Carlsson PO. Increased circulating betatrophin concentrations in patients with type 2 diabetes. Int J Endocrinol. 2014;2014:323407.
doi pubmed - Hu H, Sun W, Yu S, Hong X, Qian W, Tang B, Wang D, et al. Increased circulating levels of betatrophin in newly diagnosed type 2 diabetic patients. Diabetes Care. 2014;37(10):2718-2722.
doi pubmed - Chen X, Lu P, He W, Zhang J, Liu L, Yang Y, Liu Z, et al. Circulating betatrophin levels are increased in patients with type 2 diabetes and associated with insulin resistance. J Clin Endocrinol Metab. 2015;100(1):E96-100.
doi pubmed - Gomez-Ambrosi J, Pascual E, Catalan V, Rodriguez A, Ramirez B, Silva C, Gil MJ, et al. Circulating betatrophin concentrations are decreased in human obesity and type 2 diabetes. J Clin Endocrinol Metab. 2014;99(10):E2004-2009.
doi pubmed - Abu-Farha M, Abubaker J, Al-Khairi I, Cherian P, Noronha F, Hu FB, Behbehani K, et al. Higher plasma betatrophin/ANGPTL8 level in Type 2 Diabetes subjects does not correlate with blood glucose or insulin resistance. Sci Rep. 2015;5:10949.
doi pubmed - Ferrannini E, Muscelli E, Frascerra S, Baldi S, Mari A, Heise T, Broedl UC, et al. Metabolic response to sodium-glucose cotransporter 2 inhibition in type 2 diabetic patients. J Clin Invest. 2014;124(2):499-508.
doi pubmed - Takebayashi K, Hara K, Terasawa T, Naruse R, Suetsugu M, Tsuchiya T, Inukai T. Effect of canagliflozin on circulating active GLP-1 levels in patients with type 2 diabetes: a randomized trial. Endocr J. 2017 (in press).
- Takebayashi K, Hara K, Terasawa T, Naruse R, Suetsugu M, Tsuchiya T, Inukai T. Circulating SerpinB1 levels and clinical features in patients with type 2 diabetes. BMJ Open Diabetes Res Care. 2016;4(1):e000274.
doi pubmed - Treatment guide for diabetes edited by Japan Diabetes Society 2007 (Bunkodo Co., Ltd, Tokyo, Japan).
- Product insert: Phoenix Pharmaceuticals Inc., Burlingama, CA. Betatrophin (human) EIA Kit (catalog no. EK-051-60).
- Espes D, Martinell M, Liljeback H, Carlsson PO. Betatrophin in diabetes mellitus: the epidemiological evidence in humans. Curr Diab Rep. 2015;15(12):104.
doi pubmed - Gomez-Ambrosi J, Pascual-Corrales E, Catalan V, Rodriguez A, Ramirez B, Romero S, Vila N, et al. Altered concentrations in dyslipidemia evidence a role for ANGPTL8/Betatrophin in lipid metabolism in humans. J Clin Endocrinol Metab. 2016;101(10):3803-3811.
doi pubmed - Farbstein D, Levy AP. HDL dysfunction in diabetes: causes and possible treatments. Expert Rev Cardiovasc Ther. 2012;10(3):353-361.
doi pubmed
This article is distributed under the terms of the Creative Commons Attribution Non-Commercial 4.0 International License, which permits unrestricted non-commercial use, distribution, and reproduction in any medium, provided the original work is properly cited.
Journal of Clinical Medicine Research is published by Elmer Press Inc.